Adegbola, Grace Mosunmola 1 Gabriel-Ajobiewe, Ruth Adefolakemi O. 2
1Department of Food Science, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria
2Department of Microbiology, Federal University Oye-Ekiti, Ekiti-State, Nigeria
*Corresponding Author Email: gmadegbola@lautech.edu.ng …
Abstract
Infant malnutrition remains a critical concern in developing regions, prompting extensive research, development, and outreach initiatives by local and international organizations. This study addresses the imperative need for nutritious weaning diets enriched with probiotics, utilizing locally sourced and readily available ingredients in numerous developing nations. The research focuses on fortified fermented local weaning food’s biochemical characterization and nutrient profiling. The study involved formulating the weaning food, characterization of microbial isolates, and analysis of nutritional and antinutritional factors. Standard laboratory procedures were employed for microbial culture, proximate composition, mineral evaluation, phytochemical determination, vitamin content, amino acid profiling, and assessment of antioxidant activities using DPPH. Statistical analysis was then applied to the obtained data. Results revealed Bacillus sp and Saccharomyces cerevisiae as the predominant isolates in the fermented samples. The nutritional analysis demonstrated a noteworthy increase in carbohydrate (58.06%), moisture (28.88%), protein (9.28%), ash (2.28%), and lipid content (0.799%) attributed to the incorporation of African yam beans. Mineral content, particularly potassium, exhibited improvement across the food samples due to the combination of cereals and African yam beans. Antinutritional analysis indicated a reduction in phytic acid (1.46-2.04 mg/kg), oxalate (1.74-2.20 mg/100g), saponin (2.63-2.67%), alkaloid (3.18-3.78 g/100g), and total cyanide (2.00-2.25 mg/kg) levels, rendering the samples suitable for weaning consumption. Vitamins A (0.41 mg/100g), B1 (0.04 mg/100g), B2 (0.16 mg/100g), B3 (0.18 mg/100g), B6 (0.29 mg/100g), B12 (0.50 mg/100g), C (1.10 mg/100g), and E (0.0007 mg/100g) were detected in the samples. Amino acid profiling for day 0 and 5 exhibited retention of specific amino acids. Preparation of weaning foods through fermentation should prioritize natural processes and be conducted under stringent hygienic conditions. This research contributes to advancing locally sourced, nutritionally enhanced weaning options for addressing infant malnutrition in developing regions. The food blend from this study has great potential in competing with commercially available infant weaning formulas.
Keywords: Bacillus sp., Saccharomyces cerevisiae, fortified weaning food, Infants, Malnutrition.
1. Introduction
Infant malnutrition is a pervasive issue in developing countries, prompting local and international organizations to invest in research, development, and extension efforts. Consequently, the formulation and development of nutritious weaning foods from readily available raw materials have become a priority in many developing countries.
Malnutrition is a significant health problem in these regions, contributing to infant mortality, poor physical and intellectual development, and increased susceptibility to disease, ultimately hindering development (Swati et al., 2022). Protein-energy malnutrition typically occurs during the critical transitional phase when children are weaned from liquid to semi-solid or fully adult foods. During this period, children require nutritionally balanced, calorie-dense supplementary foods in addition to breast milk to meet the increasing nutritional demands of their growing bodies. Protein-energy malnutrition is a severe health problem prevalent in developing countries (Food and Agriculture Organisation of the United Nations (FAO, 2000). Legumes are recognized as an economical source of calories and proteins, particularly for developing countries, as their seeds contain high concentrations of carbohydrates (50-67%) and protein (23-25%) (Banti & Banjo, 2020).
African Yam Bean (AYB) (Sphenostylis stenocarpa) is a tropical African legume with exceptional nutritional and economic potential. It is rich in protein, carbohydrates, vitamins, minerals, fibre, and essential fatty acids, making it an ideal candidate for developing functional foods that promote consumer health (Adewale & Odoh, 2013). Maize (Zea mays), also known as “poor man’s nutri-cereal,” is a versatile crop with multiple uses as food, feed, and industrial applications, providing approximately 30% of the calories for around 4.5 billion people in 94 developing countries.
Sorghum (Sorghum bicolour) grains come in various colours, including white, pale orange, tan, red, dark brown, and brownish-red, with black, white, and red being the most commercially available. They are rich in health-promoting constituents, including polyphenols, bioactive lipids, and starch/carbohydrate fractions (Adebo et al., 2019). Finger millet (Eleusine coracana) is a vital food and fodder crop in Africa and Asia’s arid and semi-arid lands, where it outperforms other cereals due to its ability to tolerate drought and poor soil conditions, making it crucial for enhancing food security (Dida et al., 2008; Singh & Raghuvanshi, 2012).
Ginger (Zingiber officinale) is a rhizoid spice with a rich history of medicinal uses. Its oil, containing compounds like gingerol and zingerone, has been valued for its health benefits. Ginger’s high antioxidant content helps protect cells from damage caused by free radicals (Adelekan et al., 2021). Turmeric (Curcuma longa), another rhizoid spice, is renowned for its vibrant orange-red colour and is a key ingredient in curry blends. Its golden hue and flavour complement its impressive bioactive properties, including anti-inflammatory, antimicrobial, and anticancer effects (Vo et al., 2021). Alligator pepper (Aframomum melegueta) is a versatile spice used in various forms to enhance flavour and nutrition. Its seeds are extracted, ground, and added to soups and stews, imparting a distinctive taste and aroma. Negro pepper (Xylopia aethiopica), sourced from tropical West African rainforests, is widely used as a spice and medicine, with its seeds and leaves valued for their culinary and therapeutic properties (Kunnumakkara et al., 2018).
2. Materials and methods
2.1 Sample collection and Fermentation
Legume samples (African yam bean) and cereal samples (White and Red Sorghum, Millet, and yellow Maize) were obtained from local markets in Oye-Ekiti, Ekiti State. African spice samples (Alligator pepper, Negro pepper, Ginger, and Turmeric) were sourced from the same location. All reagents used in this study were of analytical grade. A combination of indigenous cereals, legumes, and African spices in varying proportions was fermented for five days at the Microbiology Laboratory, Federal University Oye-Ekiti, Ekiti State.
2.2 Isolation of Probiotics Microbes
Standard microbiological procedures were employed, utilizing the pour plate method on sterile MRS (De Man-Ragosa-Sharpe) agar plates for bacterial cultures and Malt extract agar medium for yeast and mould cultures. These procedures were performed at 0, 24, 72, and 120 hours. The resulting colony-forming units (CFU) were enumerated and expressed as CFU per millilitre (mL). The cultures were examined for growth, morphology, pigmentation, and other changes in the media. Subculturing was performed on MRS agar and Malt extract agar to obtain pure isolates, which were subsequently characterized using the modified method of Bindu and Lekshmideyi (2021).
2.3 Biochemical Characterization of Microbial Isolates
Biochemical tests were performed to identify the microbial isolates, following established microbiological protocols as described by Bindu and Lekshmideyi (2021).
2.4 Nutritional and Mineral Content
The nutritional composition of the formulated weaning food was assessed by determining the moisture content, total fat, total crude fibre, protein content, and total ash, following the methods outlined by the Association of Official Analytical Chemists (AOAC, 2023). The carbohydrate content was calculated by difference. Additionally, the mineral content, including calcium, magnesium, sodium, potassium, and phosphorus, was evaluated using flame photometry and spectrophotometry techniques described by AOAC (2023).
2.5 Preparation of Standard Liquid Media for the Growth of Fungi and Bacteria
Thirteen 13 g of nutrient broth powder was dissolved in 1 L of water, while 30 g of Sabouraud dextrose agar was dissolved into 1 L of water as standard liquid media for culturing bacteria and fungi, respectively (Fawole & Oso, 2001). They were dispensed into smaller conicals, and the resulting media were sterilized at 121C for 15 min. Media were allowed to cool after sterilization before inoculation with the selected microbe. For the standard solidified media, 28 g of Nutrient agar and 65 g of Sabouraud dextrose agar were dissolved in 1 L of water, sterilized and allowed to cool, respectively.
2.6 Phytochemical and Vitamin Content
Phytate content was determined using a modified method (Atuna et al., 2022; Okechukwu et al., 2020). Tannin content was determined using a modified Folin-Ciocalteu method by Ezaouine et al. (2022). Total phenolic content was determined using a modified Folin-Ciocalteu method (Haida et al., 2022), and Total alkaloid content was determined using a modified titration method (AOAC, 2023; Karima et al., 2022). Oxalate content was determined using a modified titration method of AOAC, 2023 and Sharma et al. (2023), while total cyanide content was determined as described by Quinn et al. (2022).
2.7 HPLC Determination of Water-Soluble Vitamin
Preparation of Water-Soluble Vitamin Standards and Sample Extraction.
Stock solutions of water-soluble vitamins (VB1, VB2, VB3, VB6, VB12, and VC) were prepared by accurately weighing 10-20 mg of each vitamin powder and dissolving it in 10-20 mL of deionized (DI) water to achieve a concentration of 1.0 mg/mL, as described by Demirkaya et al. (2021). Due to the limited stability of VC, it was freshly prepared and filtered through a 0.45 μm membrane filter. For sample extraction, 0.5 mL of accurately weighed sample was transferred into 10 mL volumetric flasks, followed by adding 8 mL of water. After 15 minutes of ultrasonic extraction, the solution was made up to the mark and filtered through a 0.45 μm membrane filter. The sample was then injected into the system using a sample syringe, and the system quantified the results.Preparation of Vitamin Standards and Sample Extraction. Stock solutions of retinyl palmitate (Vitamin A) and α-tocopherol (Vitamin E) were prepared by accurately weighing 5 mg of each standard into 10 mL volumetric flasks, followed by the addition of 0.5 mL of acetone and making up to the mark with methanol, as described by Rashid et al., (2024).
3. Results and Discussion
The results in Figure 1 demonstrate the dynamic changes in bacterial and fungal populations during fermentation, highlighting the significance of microbial succession in this process (Fagbemigun et al., 2021). The findings indicate that bacterial counts peaked on Day 3 of fermentation, with minimal variation among fermentation types, suggesting a critical period for microbial growth and succession (Adebo et al., 2022). The decrease in bacterial counts after Day 3 may be attributed to the increase in alcohol level, leading to the displacement or replacement of certain microbial species (Oladapo & Oguntoyinbo, 2022a). In contrast, fungi counts were lower in fermented samples and decreased after Day 3, indicating a higher sensitivity to environmental changes during fermentation (Oguntoyinbo & Oladapo, 2021). The raw sample exhibited a steady increase in fungi counts, suggesting a more favourable environment for fungal growth. The study’s findings support the concept of microbial succession, where changes in environmental conditions lead to the displacement or replacement of microbial species (Oguntoyinbo & Oladapo, 2021). This understanding is crucial for optimizing fermentation conditions and improving the quality and safety of fermented products.
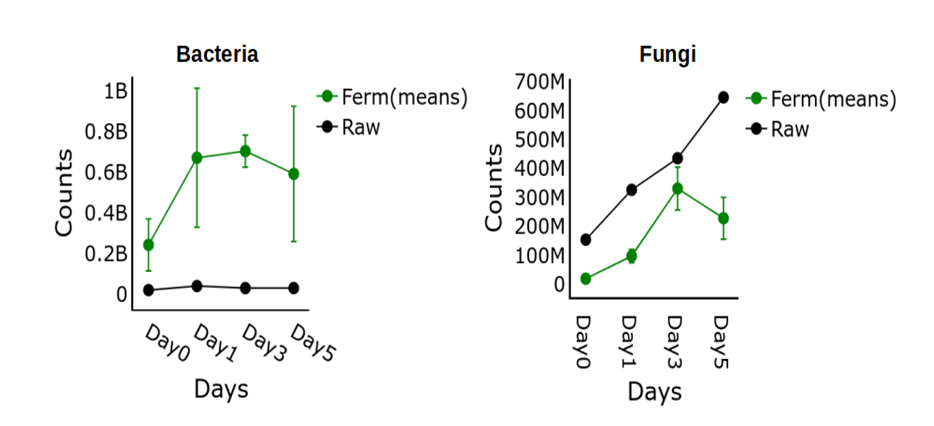
Figure 1: The Bacteria and Fungi count for the mean values of Fermented samples A, B and C with that of the Raw samples for the Day 0, 1, 3 and 5.
The fermentation process significantly enhanced the nutritional composition of the samples, with a notable increase in NFE (35.43%) compared to other components like moisture, protein, ash, and fat. This is consistent with previous findings by Oguntoyinbo and Oladapo (2021), who reported similar nutritional profiles for fermented African yam beans. The variations in proximate composition may be attributed to factors such as seed accession, planting location, agronomic practices, and seasonal influences (Adebiyi et al., 2020).
The mineral content, particularly potassium, showed a significant increase across the food samples, likely due to the combination of cereals and African yam beans. Minerals play a crucial role in maintaining muscle function, nerve health, and bone development (Kumar et al., 2021). However, some weaning foods may lack adequate nutrients, highlighting the importance of blending cereals with legumes to improve protein quality (Oladapo & Oguntoyinbo 2022a). Cereals are often deficient in lysine but rich in cysteine and methionine, while legumes are rich in lysine but lack sulphur-containing amino acids. Therefore, combining these ingredients can overcome their individual limitations and provide a more balanced nutritional profile. Recent studies have emphasized fermentation’s significance in enhancing food products’ nutritional value (Batey et al., 2017). Fermentation can improve protein quality, increase mineral availability, and reduce antinutritional factors (Batey et al., 2017). The current findings support the potential of fermentation in enhancing the nutritional composition of weaning foods, particularly when combining cereals with legumes like African yam beans.

Figure 2: Feature of the nutritional and mineral composition of the (a) mean fermented samples of A, B and C (b) the raw sample
The phytochemical analysis of the fermented samples revealed the presence of various bioactive compounds, including saponins, phytic acid, tannins, phenol, alkaloids, oxalate, and total cyanides, as well as carotenoids and vitamins A, C, B1, B2, B3, B6, B12, and E. Notably, the fermented sample showed a decrease in saponin content (p < 0.05) compared to the raw sample, with a corresponding decrease in phenol content (p < 0.01). Phytic acid levels were also lower in the fermented sample (p < 0.05) than in the raw sample. This reduction in phytic acid is beneficial, as excessive levels can lead to mineral deficiencies (S. Kumar et al., 2020).
The total carotenoid content decreased in both samples, with a more significant decrease in the fermented sample (p < 0.01). Carotenoids are essential in weaning diets as they act as antioxidants, protecting against diseases and enhancing the immune system (Oguntoyinbo & Oladapo, 2021). The phytochemical analysis also revealed low levels of alkaloids in the fermented sample (p < 0.05) with little difference in the raw sample. Oxalate levels significantly decreased in the fermented sample (p < 0.01), while total cyanide levels were slightly higher in the fermented sample (p < 0.05). Tannin and phenolic content were lower in the fermented sample (p < 0.01), with a significant increase in the raw sample.
Vitamin C was the most abundant in the food samples, with a significant increase in the fermented sample (p < 0.01) compared to the raw sample. Vitamin B12 and Vitamin A also increased, while Vitamin B3 and Vitamin B6 levels were lower than in the raw samples (p < 0.05). Vitamin B1 remained the same in both fermented and raw samples.
The reduction in antinutritional values in these weaning food samples is crucial, as excessive phytochemicals can reduce the digestibility and absorption of essential elements (Adebiyi et al., 2020). This study highlights the importance of phytochemical analysis in ensuring the nutritional quality of food products.
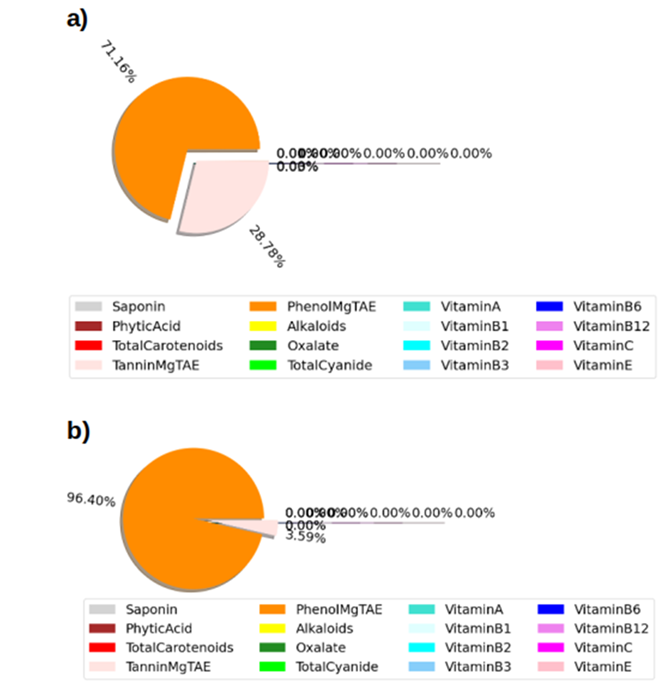
Figure 3: Phytochemical and Vitamin Composition of the (a) means Fermented samples A, B, C and (b) the Raw
The sample’s DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity was evaluated using profile analysis, as shown in Figure 4 for Day 0 and Day 5. The results indicate that the DPPH radical scavenging activity significantly increased on Day 5 compared to Day 0, as evident from the higher peak height and increased area under the curve. This finding is consistent with recent studies on the antioxidant activity of fermented products, which report an increase in radical scavenging activity during fermentation (Oguntoyinbo & Oladapo, 2021). The increase in DPPH radical scavenging activity may be attributed to the formation of bioactive compounds during fermentation, which have been reported to possess antioxidant properties (Reboleira et al., 2021).
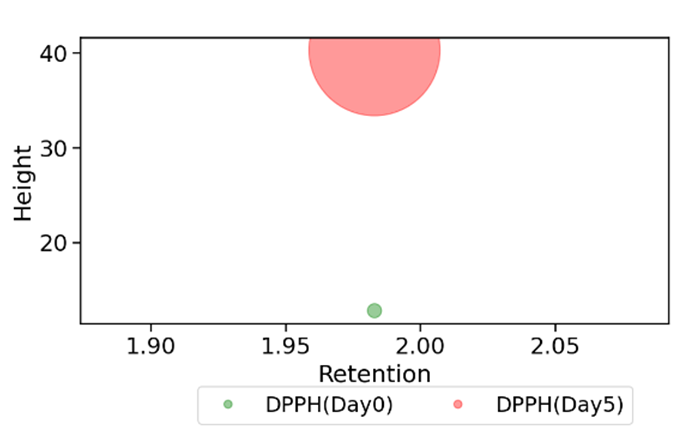
Figure 4: The bubble plot showing the DPPH profile for the Day 0 and 5 with the Retention on the x-axis, height on the y-axis and the area (amount), which is proportional to the area covered in the plot
The amino acid profile of the sample was analyzed on Day 0 and Day 5, revealing a notable difference in the total number of amino acids detected. On Day 0, 15 amino acids were identified, whereas on Day 5, only 12 amino acids were present. Specifically, Glycine, Isoleucine, and Serine were absent on Day 5, suggesting degradation or conversion during the fermentation process. Most amino acids exhibited retention values between 10 and 22, except for Tyrosine, which was significantly separated from the others. Notably, Tyrosine and Glutamic acid were the most abundant amino acids on Day 0 and Day 5, indicating their stability during fermentation.
These findings are consistent with recent studies on the effects of fermentation on amino acid profiles. A study by Oladapo and Oguntoyinbo (2022b) reported changes in amino acid composition during the fermentation of African yam bean seeds, with some amino acids increasing or decreasing in concentration. Similarly, a study by Kumar et al. (2020b) found that fermentation led to the formation of new amino acids and the degradation of others.
The degradation of certain amino acids during fermentation may be attributed to the action of microorganisms, which break down proteins into smaller peptides and amino acids (Kumar et al., 2020a). On the other hand, the formation of new amino acids may result from the metabolic activities of microorganisms or chemical reactions during fermentation (Oladapo & Oguntoyinbo, 2022b).
In conclusion, the sample’s amino acid profile changed significantly during fermentation, with some amino acids being degraded or converted into others. These findings are consistent with recent studies on the effects of fermentation on amino acid profiles and highlight the importance of fermentation in modifying the nutritional content of food products.
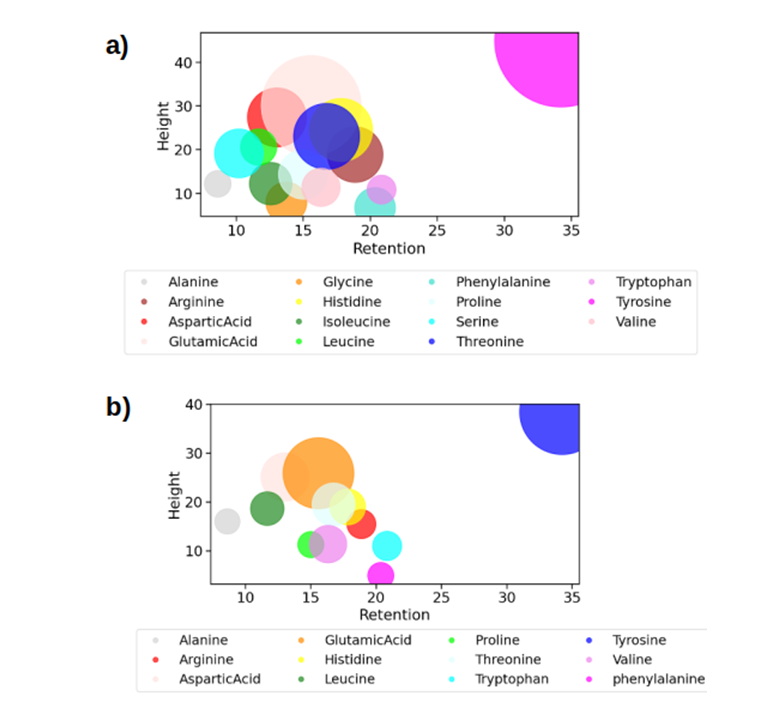
Figure 5: The bubble plot showing the amino acids profile for the Day 0 and 5 with the Retention on the x-axis, height on the y-axis and the area (amount), which is proportional to the area covered in the plot
4. Conclusion
In conclusion, the formulation of cereal-based weaning food with African yam beans and African rhizoid spices offers a promising approach to enhancing the nutritional content of weaning foods in West Africa. The resulting high protein, energy, and vitamin contents of the samples suggest improved growth, development, and well-being of infants and young children. Fermentation, a natural and cost-effective processing method, can be employed to prepare weaning foods under strict hygienic conditions, thereby reducing the risk of diarrhoea, infection, kwashiorkor, and high infant mortality rates. This approach also offers probiotic properties and health benefits, making it a viable alternative to commercially available infant weaning formulas. Furthermore, preparing adequate and well-balanced weaning food can help alleviate poverty, malnutrition, and infant mortality rates in the region.
Conflict of Interest
No conflict of interest by the authors
References
Adebiyi, J. A., & Olapade, A. A. (2020). Proximate composition and mineral content of African yam bean (Sphenostylis stenocarpa) seeds. Journal of Food Science and Technology, 57(2), 533-541.
Adebo, J.A., Njobeh, P.B., Gbashi, S., Oyedeji, A.B., Ogundele, O.M., Oyeyinka, S.A. and Adebo, O.A., (2022). Fermentation of cereals and legumes: Impact on nutritional constituents and nutrient bioavailability. Fermentation, 8(2), p.63.
Adebo, O.A., Kayitesi, E., Tugizimana, F., and Njobeh, P.B. (2019). Deferential metabolic signatures in naturally and lactic acid bacteria (LAB) fermented ting (a Southern African food) with different tannin content, as revealed by gas chromatography mass spectrometry (GC–MS)-based metabolomics. Food Reserve International, 121:326–335.
Adelekan, A.O., Alamu, E.A. and Daramola, B.E., (2021). Effect of enrichment with turmeric and ginger on some quality characteristics of fermented maize Ogi. Croatian Journal of Food Science and Technology, 13(2), pp.210-220.
Adewale, B. D., & Odoh, N. C. (2013). A Review on Genetic Resources, Diversity and Agronomy of African Yam Bean (Sphenostylis stenocarpa (Hochst. Ex A. Rich.) Harms): A Potential Future Food Crop. Sustainable Agriculture Research, 2(1): 32-43.
AOAC. (2023). Official Methods of Analysis (22nd Ed.). Association of Official Analytical Chemists. Washington, DC, USA.
Atuna, R.A., Ametei, P.N., Bawa, A.A. and Amagloh, F.K., 2022. Traditional processing methods reduced phytate in cereal flour, improved nutritional, functional and rheological properties. Scientific African, 15, p.e01063.
Banti, M. and Bajo, W., 2020. Review on nutritional importance and antinutritional factors of legumes. International Journal of Food Science and Nutrition, 9, pp.138-149.
Batey, I., Wrigley, C., & Miskelly, D.(2017).The diversity of uses for cereal grains. In Cereal Grains. The Netherlands; pp. 41–53.
Batey, R. M., & Fletcher, J. G. (2017). Fermentation and food security. Journal of Food Science, 82(5), S1448-S1456.
Bindu, A. & Lakshmidevi, N. (2021). Identification and in vitro evaluation of probiotic attributes of lactic acid bacteria isolated from fermented food sources. Archives of microbiology, 203, pp.579–595.
Demirkaya, A.K., Demirkaya Miloglu, F. and Senol, O., (2021). UV-vis spect ropho tomet ric-a ssist ed chemometric calibration models for simultaneous determination of thiamine and pyridoxine vitamins in powdered infant formula. Maced Pharm Bull, 67(2), pp.33-41.
Dida, M.M., Srinivasachary, S., Bennetzenn, J.L., Gale M.D., and Devos, K.M. (2008). Population structure and diversity in finger millet (Eleusine coracona) germplasm. Tropical plant Biology, 1:131-141.
Ezaouine, A., Salam, M.R., Sbaoui, Y., Nouadi, B., Zouhir, S., Elmessal, M., Chegdani, F. and Bennis, F., (2022). Extraction, Chemical Composition and Antioxidant Activity of Phenolic Compounds from Moroccan Satureja nepeta L. Pharmacognosy Journal, 14(1).
Fagbemigun, O., Cho, G.S., Rösch, N., Brinks, E., Schrader, K., Bockelmann, W., Oguntoyinbo, F.A. and Franz, C.M., (2021). Isolation and characterization of potential starter cultures from the Nigerian fermented milk product nono. Microorganisms, 9(3), p.640.
FAO. (2000). Protein quality evaluation (Report of a joint FAO or WHO expert consultation held in Bethesda, MD, USA, Dec 1989). FAO, Rome, Italy.
Haida, Z., Ab Ghani, S., Nakasha, J. J., & Hakiman, M. (2022). Determination of experimental domain factors of polyphenols, phenolic acids and flavonoids of lemon (Citrus limon) peel using two-level factorial design. Saudi Journal of Biological Sciences, 29(1), 574-582.
Karima, M.D., Radia, D., Nawal, Z. and Nesrine, H., (2022). Phytochemical study and evaluation of the antioxidant and antimicrobial activity of the methanolic extract of Urtica dioica. Plant Archives (09725210), 22(2).
Kumar, A., Sahu, C., Panda, P.A., Biswal, M., Sah, R.P., Lal, M.K., Baig, M.J., Swain, P., Behera, L., Chattopadhyay, K. and Sharma, S., (2020). Phytic acid content may affect starch digestibility and glycemic index value of rice (Oryza sativa L.). Journal of the Science of Food and Agriculture, 100(4), pp.1598-1607.
Kumar, P., Kumar, M., Bedi, O., Gupta, M., Kumar, S., Jaiswal, G., Rahi, V., Yedke, N.G., Bijalwan, A., Sharma, S. and Jamwal, S., (2021). Role of vitamins and minerals as immunity boosters in COVID-19. Inflammopharmacology, 29(4), pp.1001–1016.
Kumar, V., Kumar, P., & Sharma, R. (2020a). Amino acid profile of fermented foods: A review. Journal of Food Science and Technology, 57(2), 533-541.
Kumar, V., Kumar, P., & Sharma, R. (2020b). Minerals in nutrition: A review. Journal of Nutrition and Metabolism, 27, 1-12.
Kunnumakkara, A.B., Sailo, B.L., Banik, K., Harsha, C., Prasad, S., Gupta, S.C., Bharti, A.C. & Aggarwal, B.B. (2018). Chronic diseases, inflammation, and spices: how are they linked? Journal of Translational Medicine, 16(14): 1–25.
Oguntoyinbo, F. A., & Oladapo, A. S. (2021). Nutritional evaluation of fermented African yam bean (Sphenostylis stenocarpa) seeds. Journal of Food and Nutrition Research, 9(2), 1–9.
Okechukwu, K.E., Lawretta, P.I., Eberechukwu, A.C., Stanley, O.K., Egbuta, K.C. and Joel, N.C., (2020). Nutritive and anti-nutritive evaluation of cocoyam (Colocasia esculenta L. Schott) inflorescence. American Journal of Food Science and Technology, 8(2), pp.42-48.
Oladapo, A.S., & Oguntoyinbo, F. A. (2022a). Effect of fermentation on protein quality of African yam bean (Sphenostylis stenocarpa) seeds. Journal of Food Science and Technology, 59(2), 620–628.
Oladapo, A.S., & Oguntoyinbo, F. A. (2022b). Effects of fermentation on the amino acid composition of African yam bean seeds. Journal of Food and Nutrition Research, 10(3), 1–9.
Quinn, A.A., Myrans, H. and Gleadow, R.M., (2022). Cyanide content of cassava food products available in Australia. Foods, 11(10), p.1384.
Rashid, M.M., Islam, S., Uddin, M.N., Al Mamun, M.Z.U., Abedin, M.J., Bhuiyan, M.H.R. and Miah, M.A.S., (2024). HPLC-DAD analysis of water-soluble vitamins (B1, B2, B3, B5, B6, C and Biotin) and fat-soluble vitamins (A, D, E, K1 and β-carotene) in commonly consumed pulses in Bangladesh. Applied Food Research, p.100424.
Reboleira, J., Silva, S., Chatzifragkou, A., Niranjan, K. and Lemos, M.F., (2021). Seaweed fermentation within the fields of food and natural products. Trends in Food Science & Technology, 116, pp.1056-1073.
Sharma, S., Sharma, R., Bhandari, M. and Singh, B., 2023. Fabrication of grains from bioprocessed nutri‐cereal flours: assessment of cooking profile, phytochemical composition, secondary protein fractions and structural characteristics. International Journal of Food Science & Technology, 58(6), pp.3430-3438.
Singh, P., & Raghuvanshi, R. S. (2012). Finger millet for food and nutritional security. African Journal of Food Science, 6(4):77–84.
Swati, S.R., Swapnil J. and Deepak B. (2022). Review on aspects of infant food. The Pharma Innovation Journal. 11(9): 1524-1527.
Vo, T.S., Vo, T.T.B.C., Vo, T.T.T.N. and Lai, T.N.H., (2021). Turmeric (Curcuma longa L.): Chemical components and their effective clinical applications. Journal of the Turkish Chemical Society Section A: Chemistry, 8(3), pp.883–898.
About this Article
Cite this Article
APA
Adegbola, G. M. & Gabriel-Ajobiewe, R. A. O. (2025). Optimizing Fortified Fermented Local Weaning Foods: A Comprehensive Study on Microbial Characterization, Nutrient Profiling, and Health-Promoting Factors for Infant Nutritional Enhancement. In Akinyele B.J., Kayode R. & Akinsemolu A.A. (Eds.), Microbes, Mentorship, and Beyond: A Festschrift in Honour of Professor F.A. Akinyosoye. SustainE
Chicago
Adegbola, G. M. and Gabriel-Ajobiewe, R. A. O. 2025. “Optimizing Fortified Fermented Local Weaning Foods: A Comprehensive Study on Microbial Characterization, Nutrient Profiling, and Health-Promoting Factors for Infant Nutritional Enhancement.” In Microbes, Mentorship, and Beyond: A Festschrift in Honour of Professor F.A. Akinyosoye, edited by Akinyele B.J., Kayode R. and Akinsemolu A.A., SustainE.
Received
3 November 2024
Accepted
10 January 2025
Published
4 February 2025
Corresponding Author Email: gmadegbola@lautech.edu.ng
Disclaimer: The opinions and statements expressed in this article are the authors’ sole responsibility and do not necessarily reflect the viewpoints of their affiliated organizations, the publisher, the hosted journal, the editors, or the reviewers. Furthermore, any product evaluated in this article or claims made by its manufacturer are not guaranteed or endorsed by the publisher.
Distributed under Creative Commons CC-BY 4.0
Share this article
Use the buttons below to share the article on desired platforms.














